A pig kidney has now functioned inside a living human for over two months, the longest such transplant has ever lasted. The recipient, a 54-year-old woman with end-stage renal disease, is living at home, off dialysis, with a genetically modified organ from an animal. What seemed like science fiction a decade ago is becoming clinical reality.
This milestone represents the culmination of decades of research into xenotransplantation, the transplanting of organs across species. The barrier was always rejection: the human immune system recognizes pig tissue as foreign and attacks it viciously. Gene editing has changed that equation. By modifying pig DNA to remove certain markers and add human-compatible proteins, scientists have created organs that human bodies can tolerate.
For the more than 100,000 Americans currently waiting for organ transplants, and the 17 who die each day while waiting, this isn't an abstract scientific achievement. It's the beginning of a potential solution to a shortage that has killed hundreds of thousands over the past three decades.
How Gene Editing Solved the Rejection Problem
The human immune system evolved to distinguish self from non-self, and it's extremely good at its job. Pig cells carry surface proteins, particularly one called alpha-gal, that trigger immediate and violent immune rejection in humans. Previous xenotransplantation attempts failed within hours or days as the recipient's body destroyed the foreign organ.
CRISPR gene editing changed what was possible, part of a broader wave of breakthroughs in gene-editing technology. Scientists can now precisely knock out the genes that produce rejection-triggering proteins, making the pig organ less visible to human immune radar. They can also insert human genes that produce proteins the immune system recognizes as "self," further camouflaging the organ.
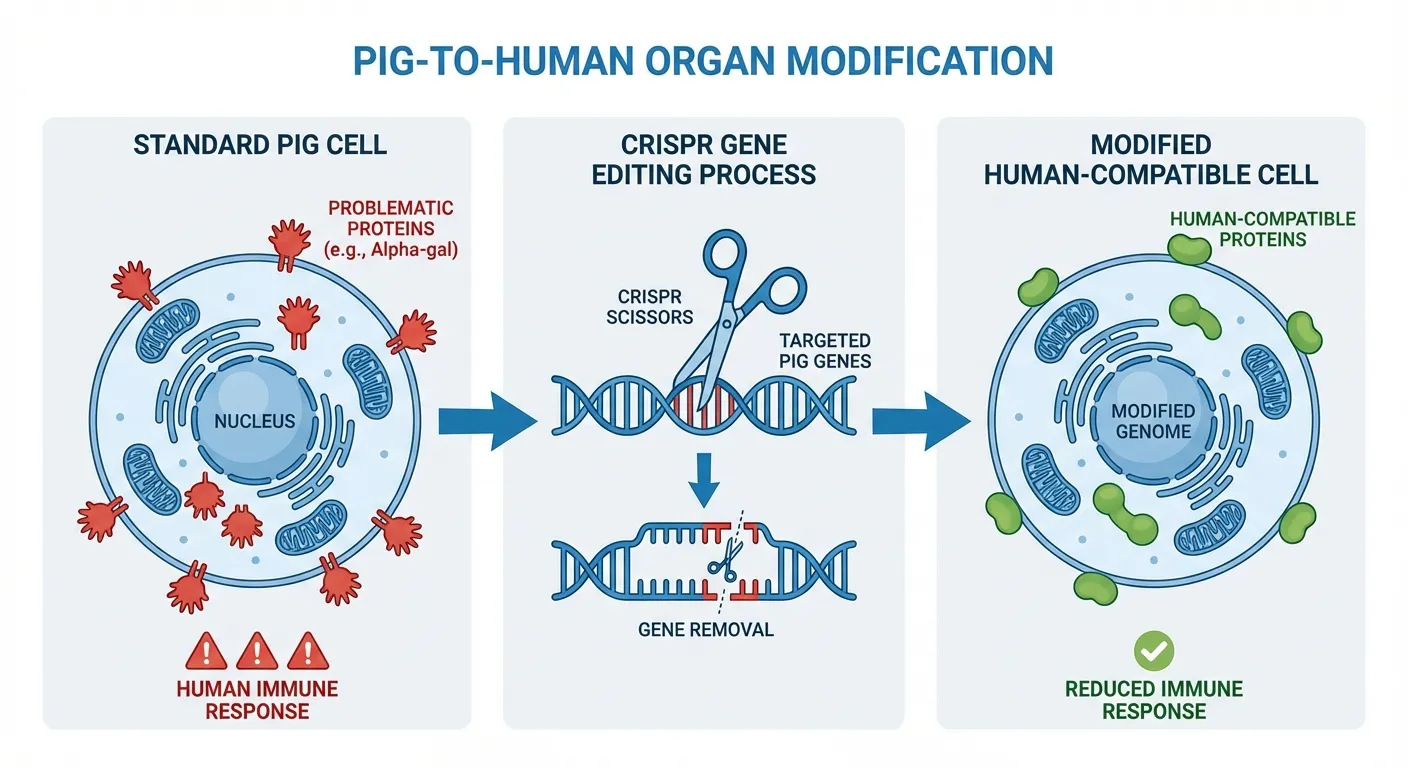
The current generation of transplant pigs has approximately ten genetic modifications. Three genes that trigger rejection have been knocked out. Six human genes have been added to improve compatibility and regulate blood clotting. One pig gene that causes organs to grow too large has been inactivated. These aren't random edits; each modification addresses a specific failure mode identified in earlier experiments.
Dr. Robert Montgomery, chair of the Department of Surgery at NYU Grossman School of Medicine and director of the NYU Langone Transplant Institute, has performed more xenotransplant surgeries in humans than any other surgeon and calls the latest results "a turning point" for patients who would otherwise die waiting. The company leading this work, eGenesis, raises pigs in biosecure facilities designed to minimize the risk of infectious disease transmission. Pigs are natural reservoirs for viruses that could potentially infect humans, so the animals are bred and raised in conditions approaching those of laboratory mice, with constant monitoring and testing.
What This Means for the Transplant Waiting List
The numbers are staggering. About 90,000 Americans are waiting for a kidney transplant right now. Only about 25,000 kidney transplants happen each year, and many of those go to patients whose conditions deteriorated enough to push them up the priority list. The median wait time for a kidney from a deceased donor is three to five years, depending on blood type and location.
Dialysis, the alternative to transplant, is a brutal existence. Patients typically spend four hours, three times per week, connected to a machine that filters their blood. Quality of life plummets. Life expectancy on dialysis is significantly shorter than with a functioning transplant. And dialysis costs the healthcare system about $90,000 per patient per year, far more than the long-term cost of a successful transplant.
If pig kidneys can be produced reliably, the entire calculus changes. Pigs breed quickly and can be raised at scale. The supply of organs would no longer be constrained by the tragedy of human death. Patients could receive transplants before years of dialysis damage their bodies. The economics would shift dramatically in favor of transplantation.
Researchers estimate that xenotransplantation could eventually provide enough organs to eliminate the kidney waiting list entirely. That's not happening tomorrow, but it's no longer a distant dream either. Clinical trials are expanding, and Mike Curtis, president and CEO of eGenesis, has stated the company's goal is to treat over 20 patients in the next couple of years. Some experts predict FDA approval for pig kidney transplants within five years if current results hold.
The Challenges That Remain
Two months is a milestone, but it's not a cure. Human kidney transplants from deceased donors typically last ten to fifteen years; transplants from living donors often last longer. Whether pig kidneys can approach that durability remains unknown. The patients who have received pig organs so far are essentially living experiments, providing data that can't be gathered any other way.
Long-term immune suppression remains a concern. Recipients of any transplanted organ must take immunosuppressive drugs to prevent rejection, and these drugs carry significant side effects, including increased susceptibility to infections and cancer. It's not yet clear whether pig organ recipients will need more or less immunosuppression than those with human organs, or whether the risks will be similar.
There's also the question of infection. Pig genomes contain endogenous retroviruses, viral DNA that's integrated into pig cells and passed from generation to generation. While these viruses don't cause disease in pigs, their behavior in human bodies over years is unknown. Gene editing can inactivate many of these viral sequences, and current transplant pigs have been extensively deactivated, but uncertainty remains.
The ethical questions are complex. Animal rights advocates oppose raising pigs for organ harvesting, viewing it as an extension of factory farming into the medical sphere. Others argue that saving human lives justifies the practice, particularly since pigs are already raised and slaughtered for food at massive scale. Religious considerations also arise: some faiths prohibit pig products, creating potential dilemmas for adherents who need transplants.
The Bigger Picture
Xenotransplantation represents one of several approaches that could reshape organ transplantation in the coming decades. Artificial organs, grown-to-order organs from stem cells, and improved preservation techniques that could extend the viability of donated organs are all advancing. The field is moving on multiple fronts simultaneously.
What makes pig organs compelling is their relative simplicity compared to alternatives. Growing a kidney from scratch using a patient's own stem cells remains largely theoretical. Building a fully functional artificial kidney small enough to implant is an engineering challenge not close to solution. Pig kidneys exist; they just needed to be made compatible. Gene editing accomplished that compatibility faster than most researchers expected.
The broader implications extend beyond kidneys, and the evolving CDC guidelines on medical procedures may eventually need to account for this new category of transplant recipients. Hearts, livers, and lungs from gene-edited pigs are all being tested. A pig heart was transplanted into a human patient in 2022, functioning for two months before the patient died of causes not entirely understood. Each organ presents different challenges, but the fundamental approach, gene editing for compatibility, applies across all of them.
Where Things Stand
Xenotransplantation is the furthest along of several competing approaches, but it is not the only one. Fully artificial kidneys remain an engineering challenge: the most advanced prototype, from the Kidney Project at UCSF, is a biohybrid device roughly the size of a coffee cup that aims to begin human trials by 2027, though a fully implantable version capable of replacing dialysis is likely a decade or more away. Stem-cell-grown kidneys represent another path, with Japanese researchers at Kyoto University successfully growing functional kidney tissue in animal models, but growing a complete, transplant-ready human kidney from a patient's own cells remains years from clinical reality. Pig kidneys have the advantage of existing now, at a scale that can be expanded relatively quickly, while these alternatives continue to mature. The next few years of clinical data will determine whether xenotransplantation fulfills its promise or whether one of these competing approaches overtakes it.
Sources
- FDA greenlights trial of gene-edited pig kidneys as treatment for end-stage kidney disease - STAT News, September 2025
- First Gene-Edited Pig Kidney Transplant Clinical Trial Begins at NYU Langone Health - NYU Langone News, 2025
- Pig kidney transplants take a step forward with approval of human trials - CNN, September 2025
- eGenesis Announces IND Clearance for EGEN-2784 - eGenesis, 2025